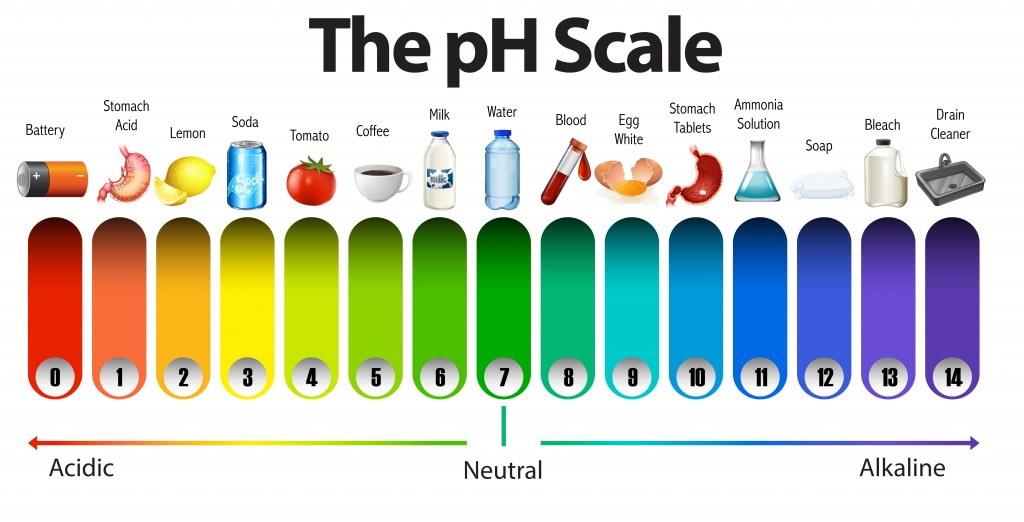
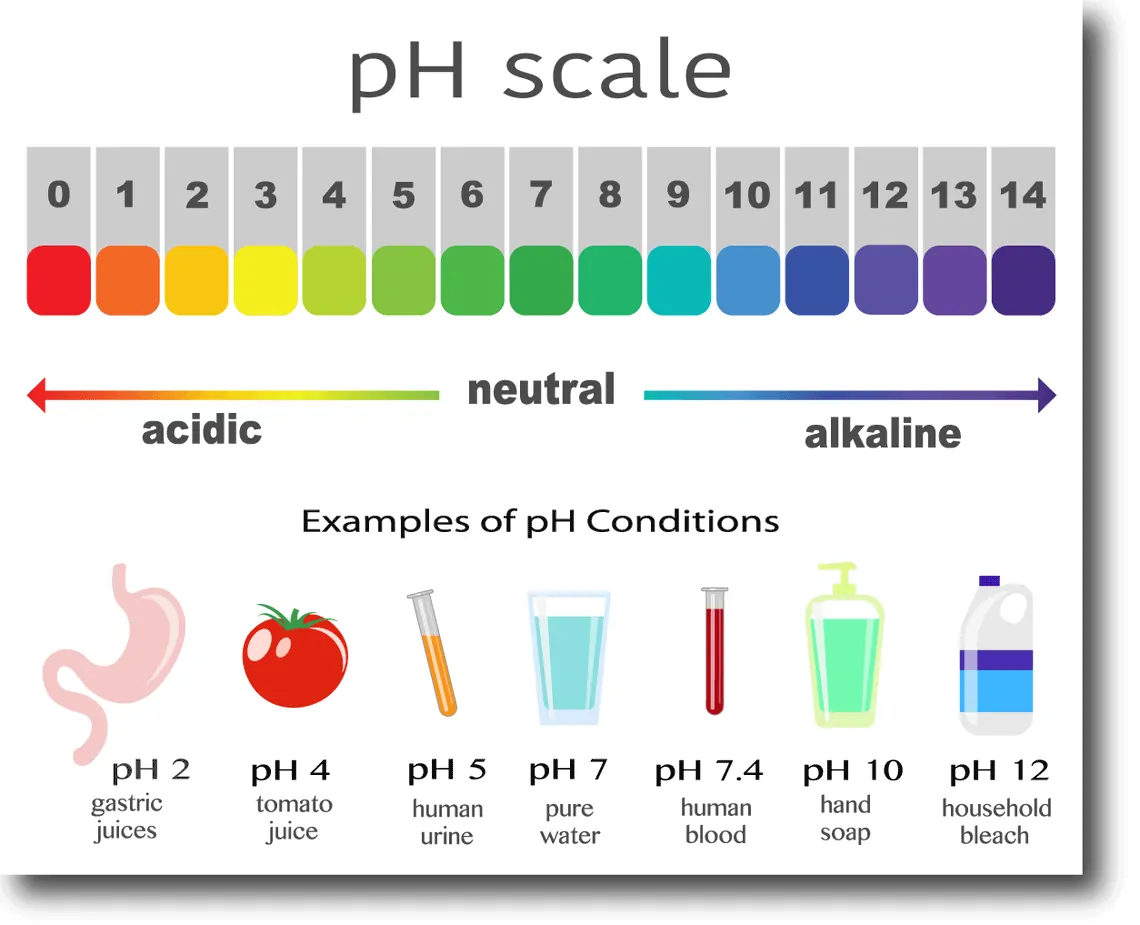
5.4 pH and pOH
4.8
(428)
Write Review
More
$ 31.00
In stock
Description
5.4 pH and pOH | Chemistry

Solved A solution has a pH of 5.4. What is its pOH? 0 -8.6
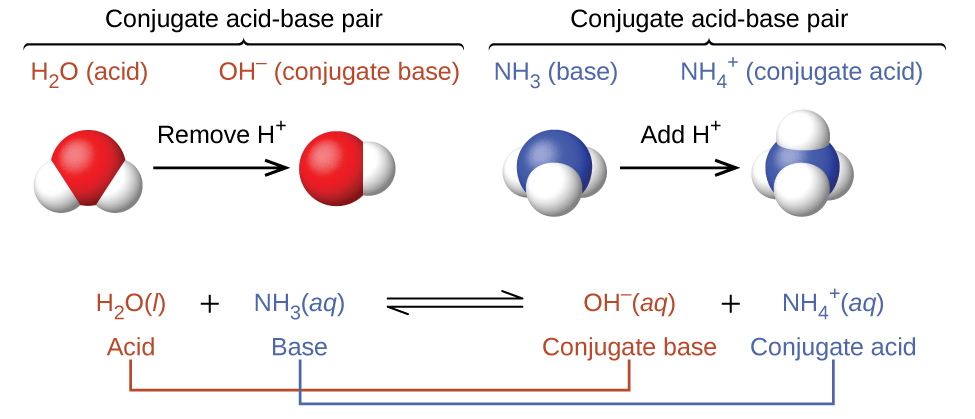
5.3: Brønsted-Lowry Acids and Bases - Chemistry LibreTexts
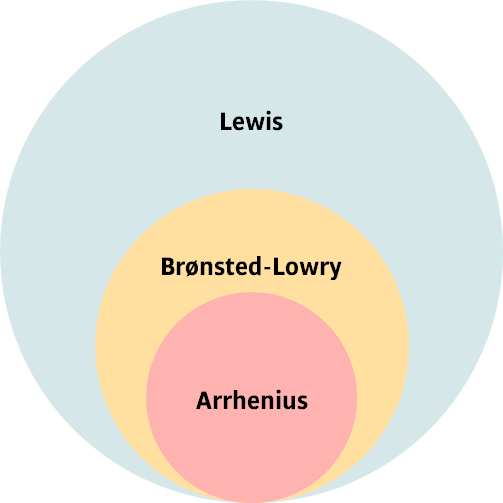
Chapter 5 Acid-Base Equilibria

AP Chemistry Workbook 2 - Edvantage Science COPIES OF THIS BOOK MAY BE OBTAINED BY CONTACTING: - Studocu

5.4 pH and pOH
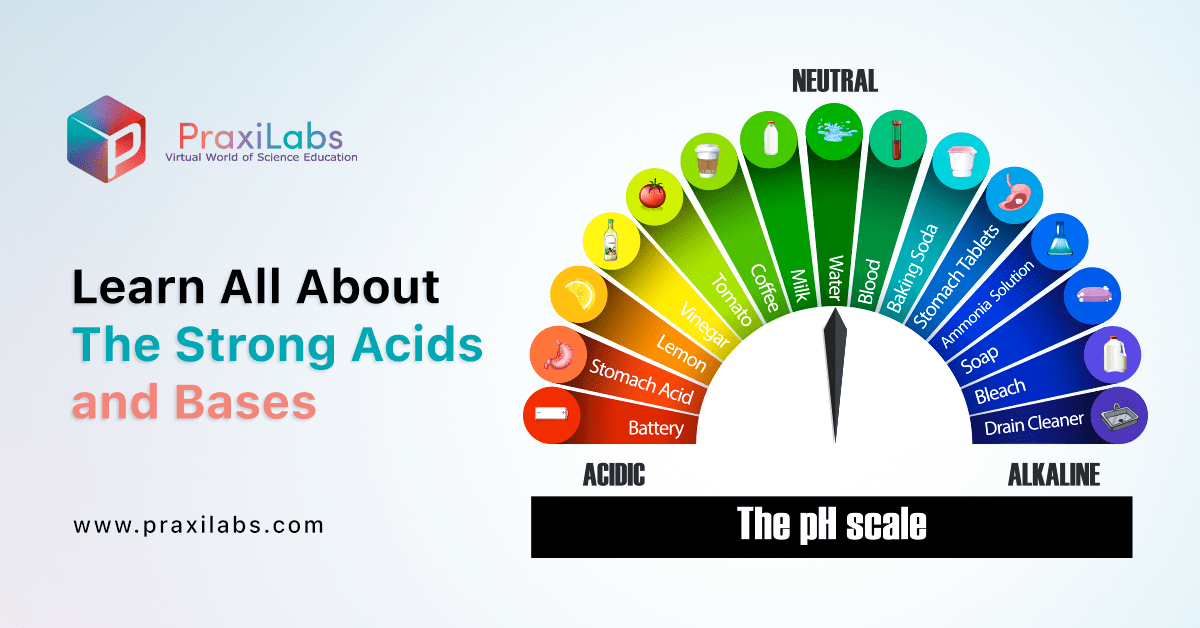
Learn All About The Strong Acids and Bases - PraxiLabs
B.2 Properties
B.2 Properties

3.7 Collision Theory

Telugu] The pH of a solution is 8.6. Calculate the OH^(-) ion concent
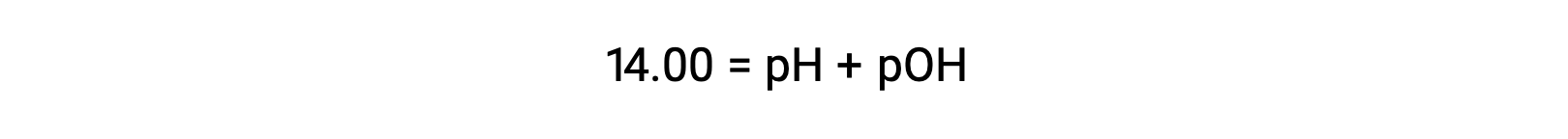
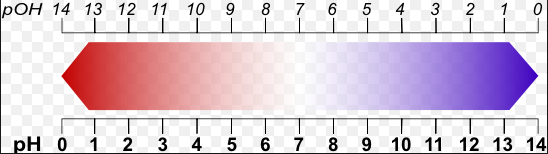
pH and pOH Scales, Chemistry
pH/pOH Concepts and Calculations - All you need to know about
You may also like