
Photograph, Titration of sodium hydroxide with hyd
License this stock photo and more images of Stock photo Titration of sodium hydroxide with hydrochloric acid. In this reaction sodium hydroxide (NaOH) is neutralized by hydrochloric acid (HCl): NaOH + HCl -> NaCl + H2O. The Erlenmeyer flask contains sodium hydroxide solution of unknown concentration. Burette above it contains 0.1M solution of hydrochlori by Science Source Images. at

Solved What reaction can occur between two of the species

Titration: Practical and Calculation (NaOH and HCl)

Acid base titration stock footage. Video of naoh, phenolphthalein

NaOH + HCl - Sodium Hydroxide & Hydrochloric Acid - Net Ionic

Titrating sodium hydroxide with hydrochloric acid

Titration hi-res stock photography and images - Alamy

Titration sodium hydroxide hi-res stock photography and images - Alamy
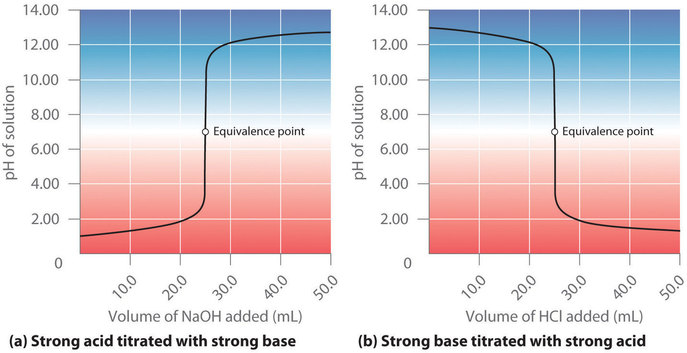
17.4: Neutralization Reactions and Titration Curves - Chemistry

NO PARKING With Specific Time Sign R7-2a Standard Traffic, 54% OFF









